As semiconductor technology races toward higher power, greater density, and miniaturization, a material with the rare combination of high thermal conductivity and excellent electrical insulation has become an industry must-have. This key member of the third-generation semiconductor family is aluminum nitride (AlN). What appears to be an ordinary white powdery ceramic has, thanks to its unique properties, quietly become indispensable in high-end fields such as 5G communications, new energy vehicles, and aerospace, driving performance breakthroughs in electronic devices.
1. Fundamental Properties of Aluminum Nitride
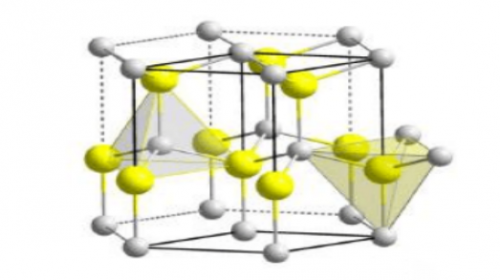
1.1 Structural Features
Aluminum nitride (AlN) is a covalently bonded compound with a hexagonal wurtzite structure. Its lattice parameters are a = 3.114 Å and c = 4.986 Å. Pure AlN appears bluish-white, but it is often gray or off-white in practice. As a typical III-V wide-bandgap semiconductor, it combines the advantages of stability and robustness.
1.2 Performance Parameters
| Parameters | Value | Notes |
|---|---|---|
| Thermal conductivity | The theoretical value is 320 W/(m·K). The actual value is greater than 180 W/(m·K). | It is 8 to 110 times that of Al2O3 |
| Coefficient of thermal expansion | 4.5 × 10⁻⁶ K⁻¹ (room temperature ~ 400°C) | Close to that of silicon (4.1 × 10⁻⁶ K⁻¹) |
| Insulation performance | Room temperature resistivity not less than 10¹⁴ Ω·cm, breakdown electric field strength 11.7 × 10⁶ V/cm | Good insulation |
| Dielectric constant | 8.8 | Comparable to Al2O3 |
| Bandgap | 6.2 eV | Higher than GaN’s 3.39 eV |
| Mechanical properties at room temperature | Hardness: 12 GPa; Elastic modulus: 314 GPa; Flexural strength: 300~400 MPa | |
| High-Temperature Mechanical Properties | At 1300℃, properties decrease by 20% (compared to room temperature) | Si3N4 and Al2O3 properties decrease by approximately 50% |
| Other | Non-toxic, excellent high-temperature corrosion resistance, decomposition temperature at normal pressure is 2000~2450℃ | BeO is toxic |
Compared to other ceramic materials, AlN exhibits outstanding overall performance, making it highly suitable for semiconductor substrates and structural packaging materials. Its potential applications in the electronics industry are vast.
1.3 Core Characteristics of Aluminum Nitride
The value of Aluminum nitride lies in its precise alignment with the physical and chemical demands of electronics, especially addressing three critical pain points: heat dissipation, electrical leakage, and signal attenuation.
- The golden combination of thermal conductivity without electrical conduction: With a room-temperature thermal conductivity of 170–200 W/(m·K)—5 to 10 times that of traditional alumina ceramics—AlN rapidly dissipates heat from electronic components, making it a “thermal champion.” At the same time, its volume resistivity reaches 10¹⁴–10¹⁶ Ω·cm, perfectly insulating against electrical current. This resolves the conflict between heat dissipation and insulation, reducing the risk of short circuits.
- High-frequency signal stabilizer: With a dielectric constant of 8.5–9.5 and dielectric loss below 0.001, AlN minimizes attenuation of high-frequency signals like 5G. At 28 GHz, signal transmittance can reach 98%.
- Extreme environment survivor: With a melting point of 2200℃ and stable performance below 1500℃, AlN is ideal for high-temperature environments. Its thermal expansion closely matches that of silicon chips, preventing component delamination due to temperature changes. It is also chemically resistant and maintains stability in humid environments.
- Non-toxic and eco-friendly: Unlike toxic beryllium oxide (BeO) ceramics, AlN is non-toxic and offers balanced performance, making it an ideal choice for electronic substrates.
2. Thermal Conductivity Mechanism
Among its many properties, AlN’s high thermal conductivity is the most remarkable. Its heat transfer primarily occurs via lattice vibrations—phonons—moving through the crystal structure. In a perfect, defect-free crystal, phonons transmit heat almost freely, giving extremely high theoretical thermal conductivity, up to 320 W/(m·K).
In reality, impurities and defects reduce thermal conductivity to below 200 W/(m·K), as uneven atomic distributions scatter phonons.

Is Aluminum Nitride Fire-Resistant?
While AlN has a high melting point of 2200℃, it is not fireproof. Similar to graphite, it reacts with oxygen above 800℃, forming aluminum oxide, which limits its use in open high-temperature environments.
Can Aluminum Nitride Melt?
At standard atmospheric pressure, AlN is a solid white to off-white powder and sublimates around 1800℃, bypassing a liquid phase. Only under extreme laboratory conditions (pressures above 10 GPa and temperatures above 2000℃) could it briefly form a liquid state. In industrial settings, Aluminum nitride behaves as a solid or dispersed nanoparticles when mixed into molten metals, rather than truly melting.
3. Applications: From Everyday Life to Cutting-Edge Technology
Thanks to its unique properties, Aluminum nitride has penetrated multiple key industries:
- Semiconductors: Aluminum nitride aln serves as the thermal core for 7nm chips. 3D-printed aluminum nitride substrates with micron-scale cooling channels can reduce chip temperatures from 120℃ to 65℃, improving thermal efficiency by 400%. It is also used in electrostatic chucks and wafer carriers, ensuring manufacturing stability.
- Optoelectronics: In blue LEDs and lasers, Aluminum nitride substrates improve optoelectronic conversion efficiency, supporting high-performance devices.
- New Energy Vehicles: AlN cooling plates in battery packs achieve over 98% contact efficiency, maintaining temperature differences within 5℃ after 1000 charge cycles. Lifespan exceeds 8 years, effectively preventing thermal runaway.
- 5G Communications: Aluminum nitride is used in base station antenna covers and filters, with insertion loss of only 0.2 dB and 60% weight reduction, ensuring signal quality and device lightness.
- Aerospace: AlN withstands -180℃ in space and up to 1200℃ during re-entry, used in navigation and control electronics, with structural precision maintained within ±0.02 mm.
- Other applications: LEDs, medical devices, and radiation-resistant nuclear components benefit from AlN’s temperature resistance, corrosion resistance, and low-loss characteristics.

4. Challenges Facing Aluminum Nitride
Aluminum nitride (AlN) still faces several challenges:
Moisture sensitivity: In humid environments, Aluminum nitride reacts with water to form aluminum hydroxide, creating an oxide layer that reduces thermal conductivity. Mitigation involves surface chemical modification or physical coating to isolate AlN from moisture.
High cost: Aluminum nitride (AlN) production requires high temperatures, leading to high energy consumption and safety risks. Impurities, carbon residues, and by-products like HCl during chemical vapor deposition require costly purification, keeping prices elevated.
5. A Promising Future: Opportunities for Innovation
As technology advances, AlN’s application scope continues to expand. Future developments include:
- Composite enhancement: Combining AlN with silicon carbide to create superior materials.
- Low-temperature sintering: Using flux agents to reduce sintering temperature from 1650℃ to below 1400℃.
- 3D printing: Developing dedicated feedstock for complex, net-shape structures.
- Emerging fields: Exploring AlN for quantum computing and advanced aerospace electronics.
With its outstanding physical and chemical properties, Aluminum nitride (AlN) is widely applied across industries. As new technologies continue to emerge, its potential grows, supporting innovation and progress in electronics and beyond.
6. Frequently Asked Questions about Aluminum Nitride
1. What is aluminium nitride used for?
Aluminum nitride (AlN) is widely used for its high thermal conductivity, electrical insulation, and chemical stability, serving in semiconductors, LEDs, lasers, EV battery cooling, 5G components, aerospace electronics, and other high-performance applications.
2. Is aluminum nitride toxic?
No, aluminum nitride (AlN) is non-toxic and safe to handle, making it a reliable alternative to hazardous ceramics like beryllium oxide.
3. Why is aluminum nitride so expensive?
Aluminum nitride (AlN) is expensive because its production requires high temperatures, strict purity control, and complex processing. Energy-intensive synthesis, removal of impurities, and safety measures all contribute to its high cost.
4. Does aluminum nitride react with water?
Yes, aluminum nitride (AlN) can react with water, forming aluminum hydroxide and releasing ammonia. This reaction can reduce its thermal conductivity, so AlN powders are often coated or chemically treated to prevent moisture damage.
